Healthcare professionals
Inspire® therapy may be a treatment option for your obstructive sleep apnea patients who are unable to benefit from CPAP.


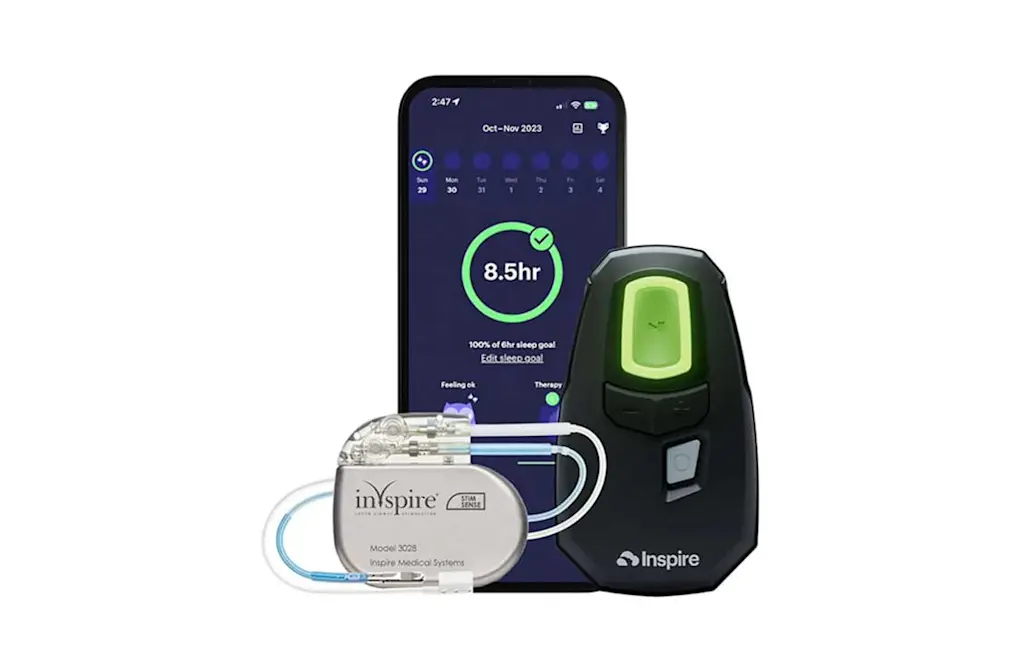
What is Inspire therapy?
Inspire therapy is a mask-free1 solution for people with obstructive sleep apnea (OSA) who have tried and struggled with CPAP. Through a simple-to-use system including the Inspire® implant, remote and app, Inspire therapy enables patients to breathe regularly so they can sleep soundly.
Many of your patients may be eligible for Inspire therapy
The FDA-approved Inspire implant is used to treat OSA in CPAP intolerant patients. Learn more about indications and contraindications for Inspire therapy.


Inspire therapy healthcare providers
See what Inspire-trained physicians are saying about their experience with Inspire therapy.
How to add Inspire therapy into your practice
It’s easy to get started—talk with an Inspire therapy expert and they’ll explain the training process. Once you’ve received your Inspire therapy training, you’ll be able to offer your CPAP-intolerant patients another option to help treat their OSA.

As an Inspire-trained physician, you’ll have all the help you could ever need.
Training modules
Our comprehensive training covers every detail.

24/7 support
We’ll always be there, whenever you need us.

Programmer demos
Practice your clinic workflow.

Educational events
Learn from those who’ve already made Inspire part of their practice.


Efficient Patient Management with the SleepSync™ Programming System
The new SleepSync Programming System enables automatic programmer uploads to SleepSync for more efficient patient management.
Commitment to quality
At Inspire Medical Systems, Inc. our focus on successful patient outcomes is paramount in everything we do. Our Quality Policy is our formal commitment to you that we will never waver in that regard.
Inspire therapy Quality Policy:
Relentlessly pursue safe, effective, and reliable treatment of obstructive sleep apnea.
Strive to consistently improve the quality of life of our patients and exceed customer expectations.
Maintain rigorous processes that ensure compliance with applicable global laws and regulations.
“Put the patient first and you will never lose your way!”
Every OSA patient experience is different, but one aspect rings true for all—it’s excruciating to live with. When it comes to OSA treatment, CPAP is the standard of care. While it works for many, it doesn’t always work for everyone. When this happens, patients feel like they’re out of options and out of luck.
From its original founding in 2007, successful patient outcomes and care experiences have been the guiding tenants for Inspire. Dr. Glen Nelson, Inspire’s first chairman of the board, established the motto, “if we put the patient first, we will never lose our way.” This spirit guides us and has led to significant investment in the Inspire post market surveillance system. Putting the patient first requires a deep understanding of the experience today and a continuous focus on identifying ways to make the experience better all the time.
Dr. Glen D. Nelson
Inspire Chairman of the Board 2007-2016
Publications
STAR 5-Year Outcomes
Evaluates and presents 5-year clinical safety and effectiveness of Inspire upper-airway stimulation for the treatment of obstructive sleep apnea.
ADHERE Registry
Updated database of “real-world” Inspire clinical outcomes.
Traditional Upper Airway Surgery VS Upper Airway Stimulation
Comparison of clinical outcomes in the treatment on obstructive sleep apnea
Safety of Traditional Sleep Surgery VS Upper Airway Stimulation
Compares readmission & complication rates 90-days postoperatively





